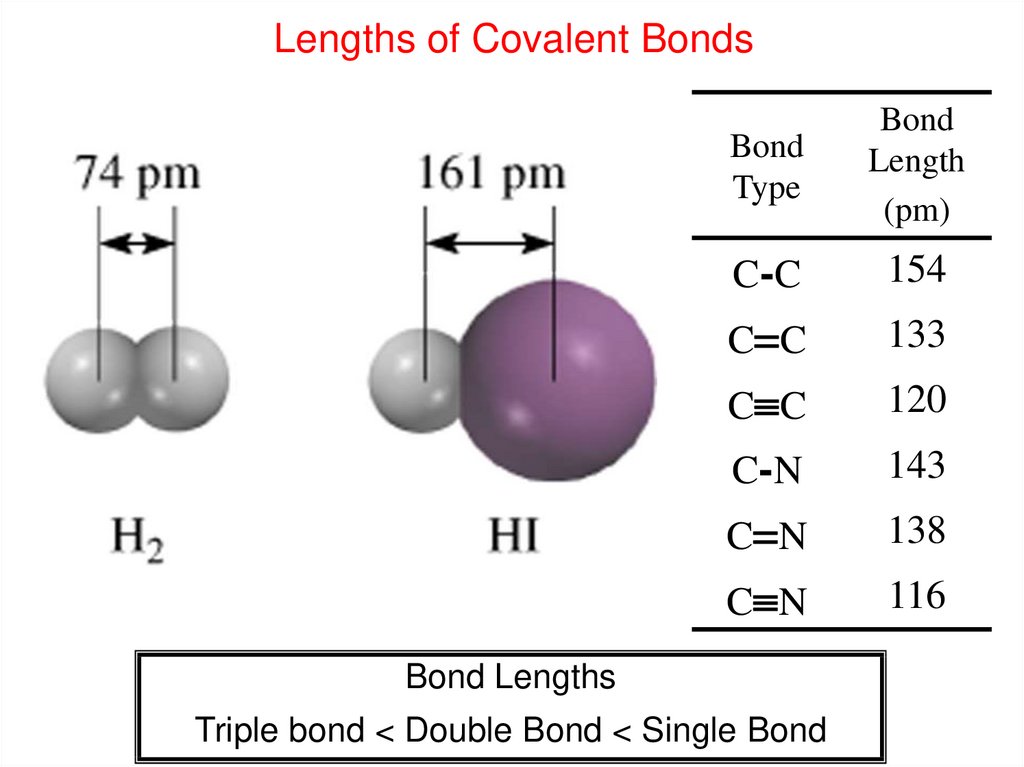 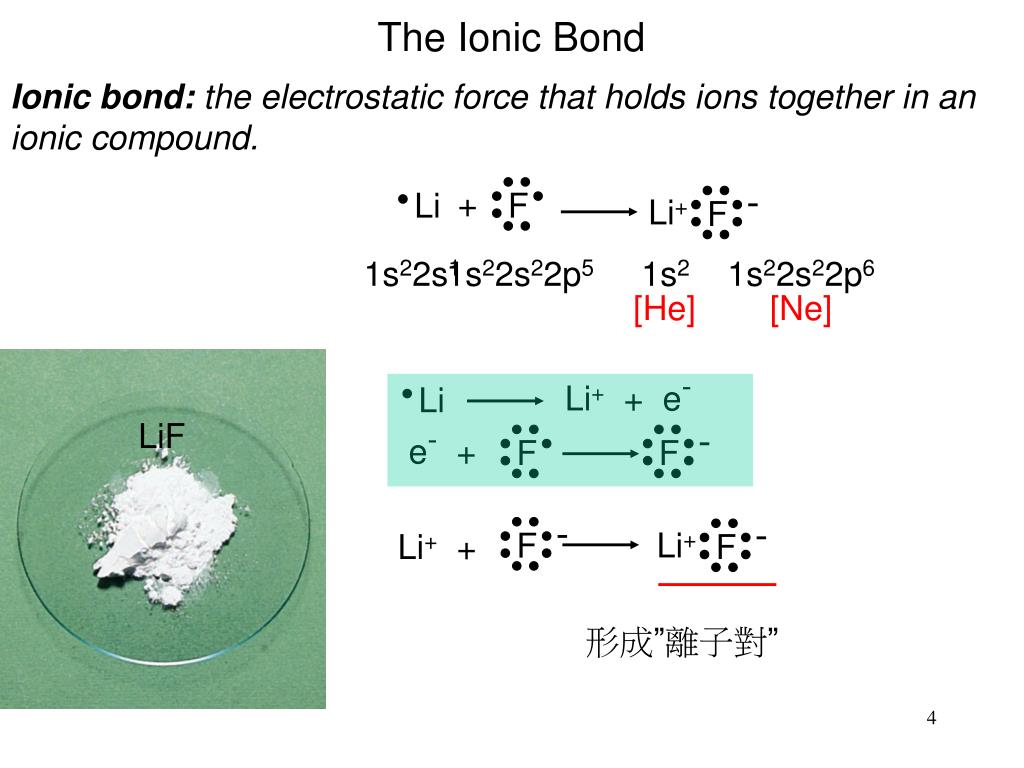 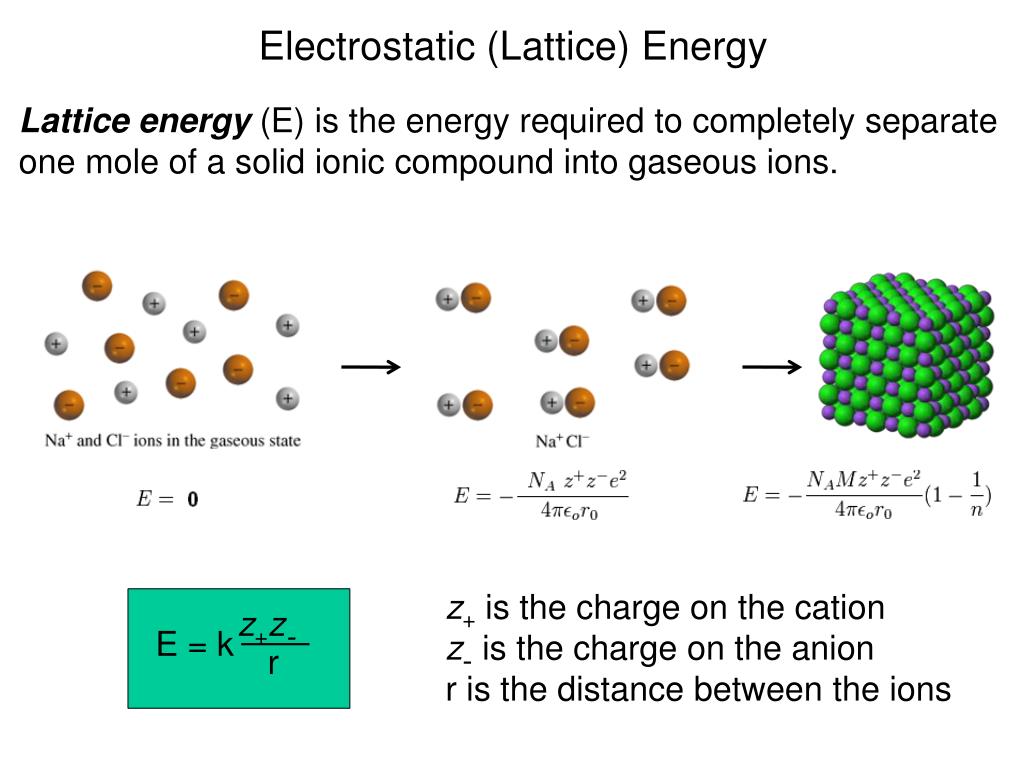
And you have learned that atoms of different elements have different numbers of protons. Remember, you have learned that there were 118 different kinds of atoms, and that each was known as an element. We're finally ready to discuss the chemical properties of the simplest chemicals out there – we're finally ready to discuss the elements. It probably seems like all we've been spending a lot of time learning about protons… and neutrons… and electrons… and electrons… and more electrons… so you might be wondering – when do we actually get to study chemistry? When do we get to study reactions? When do we get to study explosions? When do we get to study plastics, and medicines that can be made by combining different kinds of chemicals? The answer is now. 4 Group 4A-8A Continue to Add p Electrons to the Outermost Energy Level.3 Group 3A (IIIA) Elements Have s and 1 p Electrons.2 Group 2A (IIA) Elements Have Two s Electrons.1 Group 1A (IA) Elements Have One s Electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
